This research develops a physics-based method for measuring lung elasticity from medical imaging to predict which emphysema patients will benefit from lung valve treatment. By creating detailed elasticity maps, the work aims to improve treatment selection, enhance patient outcomes, and provide new quantitative tools for assessing lung health.
This research develops an AI model that combines thyroid ultrasound imaging with genetic testing to improve diagnosis of indeterminate thyroid nodules. By integrating molecular and imaging data, the model helps distinguish benign from cancerous nodules more accurately, reducing unnecessary surgeries and improving clinical decision-making for thyroid cancer patients.
This research develops soft, tissue-like implantable sensors capable of monitoring molecular signals inside the body in real time. By combining high-performance electronics with flexible, biocompatible materials, these devices could detect inflammation, stress, or organ damage before symptoms arise, enabling earlier diagnosis and more personalized healthcare.
This research investigates the genetic mechanisms underlying polycystic ovary syndrome (PCOS), a condition affecting one in ten women and the leading cause of female infertility. By studying thousands of genetic variants across multiple cell types, the project aims to identify the biological causes of PCOS and develop targeted treatments.
This research investigates macrophages, immune cells that regulate infection, tissue repair, and cancer responses. Through laboratory experiments and machine-learning models, it aims to predict macrophage function across different diseases and patients. The work could improve prognosis, guide treatments, evaluate drug safety, and forecast recovery following major illnesses and injuries.
This research develops nanobubble-enhanced ultrasound imaging as an accessible alternative to MRI for cancer diagnosis. Tiny gas-filled nanoparticles amplify ultrasound signals and improve image quality, particularly in prostate cancer. The technology could reduce diagnostic delays, lower costs, and provide high-quality medical imaging to more patients worldwide.
This research combines focused ultrasound and engineered genetic circuits to activate cancer immunotherapy directly within solid tumors. By locally triggering immune-stimulating cytokines such as IL-12, the approach aims to convert “cold” tumors into “hot” tumors while minimizing systemic toxicity, potentially expanding curative immunotherapy treatments to more cancer patients.
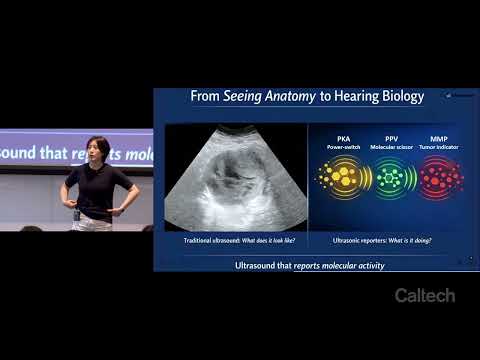
This research develops engineered ultrasonic reporters that allow ultrasound imaging to detect molecular activity rather than only anatomical structure. By targeting biological signals associated with cancer progression and cellular communication, the work aims to distinguish aggressive disease earlier and improve precision medicine through real-time, noninvasive monitoring of underlying cellular behavior.
Pagination
- Page 1
- Next page