This research develops soft, tissue-like implantable sensors capable of monitoring molecular signals inside the body in real time. By combining high-performance electronics with flexible, biocompatible materials, these devices could detect inflammation, stress, or organ damage before symptoms arise, enabling earlier diagnosis and more personalized healthcare.
This research investigates macrophages, immune cells that regulate infection, tissue repair, and cancer responses. Through laboratory experiments and machine-learning models, it aims to predict macrophage function across different diseases and patients. The work could improve prognosis, guide treatments, evaluate drug safety, and forecast recovery following major illnesses and injuries.
This research investigates how cells select which protein fragments, or peptides, to display to the immune system. Contrary to previous assumptions, peptide presentation appears highly curated rather than random. Understanding these selection rules could improve cancer immunotherapy, enhance antiviral treatments, and provide new insights into autoimmune diseases.
This research develops a noninvasive method for continuously measuring blood pressure using arterial resonance. Inspired by the physics of vibrating guitar strings, the device gently stimulates arteries and measures their resonance frequencies with ultrasound. The resulting continuous blood pressure waveforms could improve diagnosis of cardiovascular disease without invasive catheterization procedures.
This research combines focused ultrasound and engineered genetic circuits to activate cancer immunotherapy directly within solid tumors. By locally triggering immune-stimulating cytokines such as IL-12, the approach aims to convert “cold” tumors into “hot” tumors while minimizing systemic toxicity, potentially expanding curative immunotherapy treatments to more cancer patients.
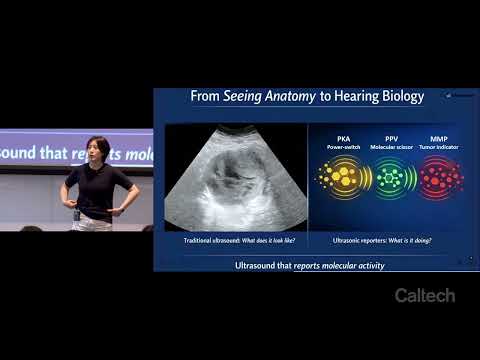
This research develops engineered ultrasonic reporters that allow ultrasound imaging to detect molecular activity rather than only anatomical structure. By targeting biological signals associated with cancer progression and cellular communication, the work aims to distinguish aggressive disease earlier and improve precision medicine through real-time, noninvasive monitoring of underlying cellular behavior.
This research investigates whether activation of the sympathetic nervous system can enhance tissue regeneration. Using engineered neural switches in mice, the study demonstrated improved healing after ear injury, including growth of nerves, blood vessels, and cartilage. The findings suggest that nervous system regulation may play an important role in future regenerative medicine therapies.
Pagination
- Page 1
- Next page