This research investigates whether activation of the sympathetic nervous system can enhance tissue regeneration. Using engineered neural switches in mice, the study demonstrated improved healing after ear injury, including growth of nerves, blood vessels, and cartilage. The findings suggest that nervous system regulation may play an important role in future regenerative medicine therapies.
This research uses artificial intelligence to predict the progression of Alzheimer’s disease and cancer using medical imaging data. By analyzing brain scans, tumor scans, and treatment responses, AI models can forecast disease development and treatment outcomes, enabling earlier intervention, more personalized care, and improved quality of life for aging populations.
This research improves photoacoustic imaging, a technique that uses light-generated sound waves to visualize tissue oxygenation deep inside the body. By calibrating measurements using highly oxygenated arterial blood, the method overcomes longstanding accuracy limitations and avoids skin-tone bias, potentially improving early tumor detection and non-invasive monitoring of tissue health.
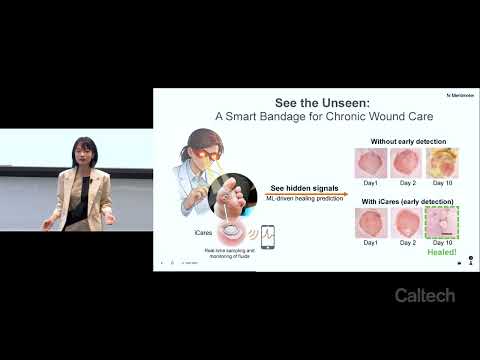
This research introduces iCares, a smart wound-monitoring bandage designed to detect infection and inflammation before visible symptoms appear. Using biosensors, fluid sampling, and machine learning, the system provides real-time wound analysis, enabling earlier intervention, personalized treatment, reduced complications, and improved healing outcomes for patients with chronic wounds.
This research models blood flow in narrowed arteries and during catheterization using the Herschel–Bulkley fluid model. By simulating flow and drug dispersion, it identifies factors affecting unpredictability. These insights enable optimized treatments, improved medical device design, and better visualization for clinicians, ultimately enhancing safety and outcomes in cardiovascular care.
This research explores asthma by recreating lung airways using 3D bioprinting. By simulating low-oxygen conditions and imaging structural changes, it investigates how exaggerated immune responses narrow airways. These models enable detailed study of disease mechanisms and offer a platform to develop treatments, ultimately advancing efforts toward preventing or curing asthma.
Pagination
- Previous page
- Page 2
- Next page