This research investigates how melanoma switches between two gene states—one fast-growing and treatable, the other slow but highly invasive and responsible for brain metastases. By identifying genes that control this transition, the study aims to force melanoma into a more treatable form, improving therapeutic options and patient outcomes.
This research investigates Large Granular Lymphocyte Leukemia, where protective T cells become cancerous. The project explores how DNA methylation silences normal T-cell function and tests drugs that reverse this process. By removing harmful chemical modifications, the goal is to restore immune cells to their healthy, protective “superhero” role.
Cancer often becomes resistant to treatment due to the protein CDK8, which helps reprogram cancer cells. Traditional inhibitors fail because CDK8 still acts as a structural scaffold. This research develops targeted degraders that use the cell’s recycling system to eliminate CDK8 entirely, preventing resistance and improving future cancer therapies.
This research targets cancer more precisely by focusing on a unique region of the PLK1 protein that drives tumor growth. By designing drugs that bind specifically to this domain using AI and laboratory testing, the approach aims to kill cancer cells while sparing healthy tissue.
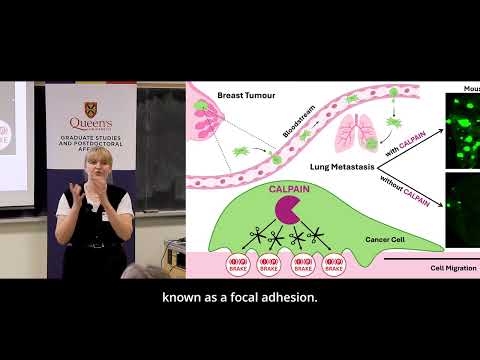
This research investigates how calpain, a protein that regulates cell movement, contributes to breast cancer metastasis. By disrupting calpain in tumor cells, researchers show reduced spread to the lungs in mouse models. Targeting calpain may improve combination therapies and offer new treatment strategies for metastatic breast cancer patients.
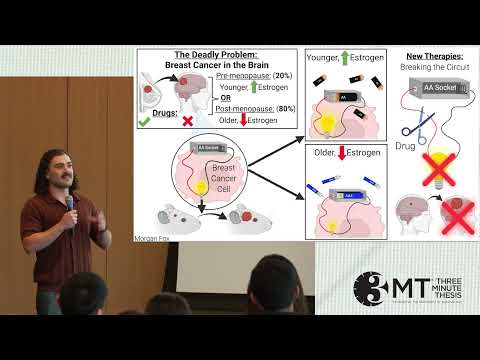
Breast cancer most often kills by spreading to the brain, where hormone therapies fail. This research reveals a signaling pathway that drives tumor growth in both pre- and postmenopausal settings. Identifying alternative activators of this pathway opens new therapeutic opportunities for deadly brain metastases.
Low-grade serous ovarian cancer frequently returns after standard treatment, and current targeted drugs eventually stop working. This research investigates why cancer cells become resistant, comparing them to prey that adapt to evade a predator. By treating patient-derived tumor cells with inhibitors and analyzing the genes activated in the resistant survivors, the research aims to uncover the mechanisms behind drug resistance and guide development of more effective therapies.
This research targets chronic lymphocytic leukemia relapse by focusing on Bruton’s Tyrosine Kinase (BTK), a key cancer-driving protein that often mutates and becomes drug-resistant. Using “molecular glues,” the project aims to degrade BTK—even when mutated—offering a new therapeutic strategy that could overcome resistance and improve outcomes for (chronic lymphocytic leukemia) CLL and other BTK-dependent cancers.
This research investigates therapy-resistant persister cells in lung cancer using single-cell analysis of patient tumors before, during, and after treatment. Findings show persister cells survive by entering a dormant state and activating the cilia–FGFR4 survival pathway. Targeting this pathway could eliminate persister cells and prevent relapse in patients.
My research develops a gentle cancer treatment using ultrasound to activate specialised LDH particles inside tumour cells. These particles release ROS “micro-explosions” that kill cancer without surgery or chemotherapy. Enhanced with MRI-visible metals, the method offers a safe, targeted therapy that destroys breast cancer cells using sound instead of pain.
Pagination
- Page 1
- Next page