This research uses spatial transcriptomics to map interactions between T cells, cancer cells, and immunosuppressive cells in tumours. Findings suggest cancer suppresses immune responses by surrounding and weakening T cells. The work aims to improve immunotherapy and enable personalised cancer treatment through detailed tumour mapping.
This research investigates how melanoma switches between two gene states—one fast-growing and treatable, the other slow but highly invasive and responsible for brain metastases. By identifying genes that control this transition, the study aims to force melanoma into a more treatable form, improving therapeutic options and patient outcomes.
This research uses yeast to study conserved molecular machinery that ensures safe chromosome division. Focusing on a key cohesin-regulating protein, the work reveals how DNA is accurately separated during cell division and how failures in this system can lead to chromosomal errors, developmental disorders, and cancer.
Mitochondria power cells and communicate with the nucleus to control gene expression. This research builds minimal artificial cells containing only mitochondria and nuclei to isolate this signaling pathway. The system reveals how mitochondrial dysfunction alters gene expression, offering new insight into mechanisms underlying cancer and neurological diseases.
This research investigates how cells repair dangerous DNA double-strand breaks through the non-homologous end joining pathway. By identifying key proteins involved in this error-prone repair process, the work reveals new opportunities to sensitise cancer cells to radiation and chemotherapy, potentially improving treatment outcomes for aggressive cancers.
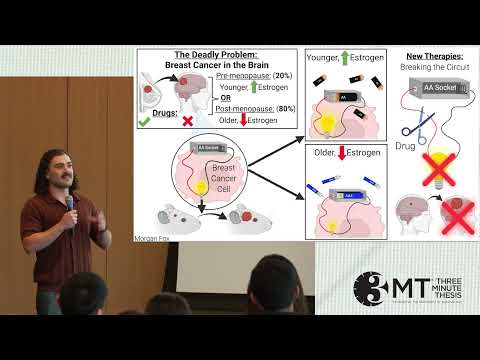
Breast cancer most often kills by spreading to the brain, where hormone therapies fail. This research reveals a signaling pathway that drives tumor growth in both pre- and postmenopausal settings. Identifying alternative activators of this pathway opens new therapeutic opportunities for deadly brain metastases.
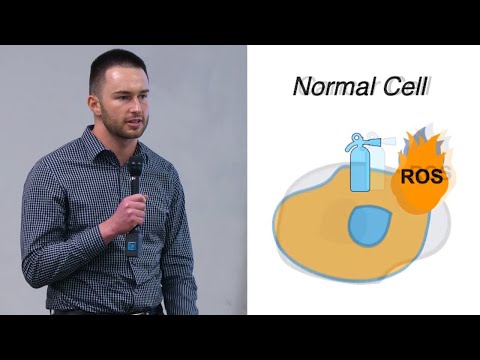
Cancer cells survive extreme oxidative stress by importing lipoproteins that deliver vitamin E, a powerful antioxidant. This creates a fire-resistant shield that prevents ferroptotic cell death. Blocking vitamin E delivery or lipoprotein uptake removes this protection, revealing a new vulnerability that could influence tumor growth and treatment response.
My research investigates collagen-binding receptors on breast cancer cells as potential biomarkers to distinguish harmless early-stage tumours from aggressive ones. Using genetically matched 3D cancer models, the project identifies how receptor activity affects invasion and collagen organization, aiming to reduce overtreatment and support clearer clinical decisions for early breast cancer patients.
My research investigates tiny particles released by metastatic cancer cells—messengers that help cancer hide from the immune system. By capturing and analysing these particles, the study aims to uncover how they evade detection and to develop new strategies that “teach” the immune system to recognise and neutralise them, leading to safer, more effective cancer therapies.
This research investigates a novel two-drug therapy for ovarian cancer that kills cancer cells without harming healthy tissues and partially reactivates the suppressed immune system. The PhD work explores how this immune “reawakening” occurs, aiming to identify new strategies to enhance it and create more effective, resistance-proof treatments.