Crimean–Congo haemorrhagic fever is a deadly tick-borne virus affecting communities in Uganda and spreading with climate change. Through interviews and large-scale antibody testing, this research identifies how people are exposed to the virus, informing targeted prevention strategies such as awareness campaigns, tick control, behavioural change, and future vaccination planning.
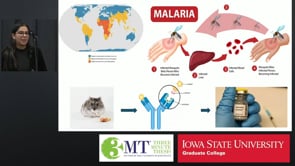
Malaria infects hundreds of millions each year by using the parasite Plasmodium to invade the liver through the CSP protein. This research designs tightly binding antibodies to block infection at its earliest stage, improving vaccine effectiveness and offering a path toward preventing malaria before symptoms begin.
By stripping Salmonella of its molecular “effectors,” this research identifies interferon gamma as a key immune barrier preventing infection. A small set of SPV genes enables the bacterium to overcome this defense. Understanding these mechanisms reveals new targets for therapies against Salmonella, a major global health threat.
Tuberculosis remains deadly despite relying on decades-old antibiotics. This research uses computational methods to identify immune response similarities between TB and other diseases, enabling drug repurposing. By borrowing already approved treatments, this approach aims to restore immune balance, combat drug resistance, and accelerate the development of new TB therapies.
This research tackles antibiotic resistance by developing nano-scale microfluidic cultures that isolate and study previously unculturable bacteria. By screening rare microbes and directly testing their antimicrobial activity, the platform accelerates discovery of new antibiotics, offering a powerful tool against drug-resistant superbugs.
A $2 portable HIV test chip that combines PCR-level sensitivity with home-test simplicity. Using magnetic microparticles, custom probes, and automated processing, it delivers rapid color-change results from a single drop of blood. The system could diagnose HIV and other viruses quickly, affordably, and anywhere.
Variants weaken current COVID vaccines because they target parts of the spike protein that mutate. This project uses nanoparticles displaying engineered versions of the conserved RBD region to steer the immune system toward making broadly protective antibodies. Computational design helps optimize immune targeting, potentially eliminating yearly boosters and protecting against future coronaviruses.
This research focuses on strengthening fragile mRNA molecules to create vaccines that are more stable, effective, and easier to distribute. By modifying mRNA structure to resist degradation, vaccines could be stored at higher temperatures and maintain potency, expanding access—especially in low-resource regions—and improving global readiness for future pandemics.
This research aims to solve the major weakness of mRNA vaccines—the need for constant cold storage—by packaging them inside ultra-stable protein “boxes” called encapsulins. These naturally robust containers protect mRNA in extreme environments. A working prototype now exists, offering the potential for globally distributable, freezer-free vaccines that remain effective anywhere.
This research uses harmless insect-specific viruses to block mosquitoes from becoming infected with dangerous human viruses like dengue or Zika. Through superinfection exclusion, an already-infected mosquito can’t host a second virus. The work explores releasing “pre-infected” mosquitoes as a safe, sustainable method to prevent disease transmission globally.
Pagination
- Previous page
- Page 2
- Next page