Myelin enables efficient communication between nerve cells and is essential for cognition, movement, and sensation. In neurodegenerative diseases, myelin is lost, impairing daily life. This research uses stem cells, gene profiling, and gene editing to uncover why myelin fails—and how regenerating it could transform treatment.
Neurodegenerative diseases like Alzheimer’s and Parkinson’s are closely linked to abnormal dopamine levels but are diagnosed too late. This research develops a tiny electrochemical brain sensor that selectively detects dopamine in real time. Such technology could enable earlier diagnosis, better monitoring, and improved treatment of neurological disorders.
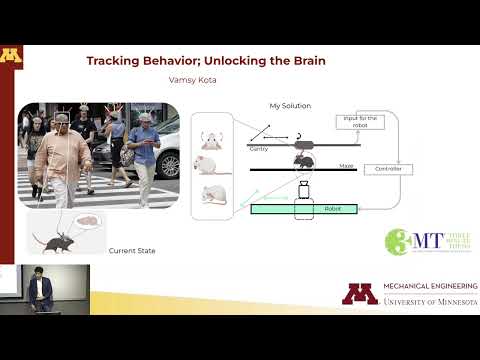
Understanding how the brain controls behavior is key to studying neurological disease. This research introduces a high-speed robotic system that tracks mouse behavior in fine detail. By synchronizing precise behavioral data with brain activity recordings, it enables researchers to link specific neural regions to actions, improving insight into disorders like Parkinson’s and Alzheimer’s.
Researchers describe a simple strategy to slow Alzheimer’s disease by capping toxic tau protein chains. Inspired by a ring-stacking toy, they engineered spiky molecular “hats” that bind tau, halt aggregation, and reduce spread in cellular and postmortem brain models, suggesting broad potential across neurodegenerative disorders with future therapeutic promise worldwide.
This research investigates how motion perception changes with age and how these changes are reflected in brain function. Using behavioural tasks and fMRI, the research aims to develop simple visual tests that could be used in routine eye-care settings to identify early signs of cognitive decline and support healthy ageing.
This study introduces a “brain stress test” for depression, combining targeted brain stimulation with neuroimaging. Depressed individuals show exaggerated brain responses, which increase with repeated episodes. The test may serve as an early warning signal, helping clinicians identify relapse risk and intervene before depression returns.
Mental health disorders disrupt neural connections in the brain, yet most treatments only manage symptoms. This research explores psychedelic-inspired drugs that restore lost brain connections without hallucinogenic effects, using automated imaging tools to identify compounds that rebuild neural structure and offer lasting recovery.
This research proposes that psychotherapy works by reshaping cognitive maps in the brain, much like navigation. In depression, these maps become narrow and repetitive. By analyzing therapy language and concept networks, this work aims to make therapy more precise—helping clinicians visualize mental “stuck points” and guide patients toward healthier paths.
Electrical signals in the body depend on ion channels that regulate salt movement across cell membranes. When these channels malfunction, diseases like epilepsy and heart arrhythmias can occur. This research decodes how faulty ion channels work, revealing potassium-based mechanisms that could restore electrical signaling and guide new therapies.
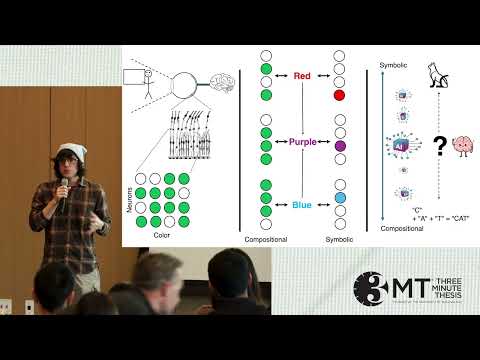
This research investigates the neural “language” of vision, asking whether the brain encodes images using compositional or symbolic patterns. Using machine learning and artificial neural networks, the work reveals evidence for a compositional visual code, informing the future design of advanced visual prosthetics.
Pagination
- Previous page
- Page 3
- Next page