This research targets rare genetic diseases caused by frameshift mutations using antisense oligonucleotides as “genetic band-aids.” By masking faulty DNA segments, it restores functional protein production. Demonstrated in muscular dystrophy models, this approach offers a scalable strategy to treat multiple rare diseases, addressing a major gap where most conditions lack effective therapies.
Genetic cardiomyopathies arise from DNA errors that disrupt vital heart proteins and can be fatal in childhood. This research improves heart-targeted gene therapy by guiding treatments through the bloodstream using chemokine “traffic signals” and avoiding immune interference, enabling therapies to reach the heart more efficiently and potentially cure inherited heart disease.
Congenital heart defects are the leading cause of infant death from birth defects. This research develops a high-throughput method to test genetic mutations in key heart genes like TBX5, identifying which variants disrupt heart development. The approach improves diagnosis, informs gene therapy, and advances understanding of why hearts fail before birth.
SLC13A5 citrate transport disorder causes severe neonatal seizures due to disrupted citrate balance in the brain. This research uses mouse models to show excess citrate worsens seizures and explores gene replacement therapy to restore transporter function. Early results show reduced seizures, with human clinical trials beginning soon.
Craniosynostosis occurs when skull sutures fuse too early, requiring risky surgeries. The researcher identified microRNA-200A as a key regulator of suture development. In mice lacking miR-200A, sutures fused prematurely, but adding extra miR-200A via gene therapy prevented fusion entirely. This breakthrough suggests a non-surgical future treatment for craniosynostosis.
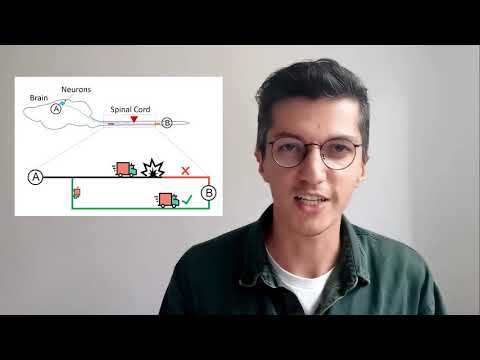
This research explores neural remodeling—the process by which neurons form new connections after spinal cord injury. Using mouse models, the work identifies genes involved in detour pathways and enhances them through gene therapy, strengthening recovery. The goal is to develop future treatments that improve functional outcomes for people with central nervous system injuries.
This research tests whether positive, therapy-induced epigenetic changes can be inherited. Using mice with genetic eye disease, the team applies a successful treatment, checks for vision improvement, examines resulting DNA chemical marks, and studies whether offspring inherit these beneficial modifications. Findings could reshape our understanding of therapy and generational health impact.